|
Vesicles located closer to Ca 2+ channel(s) are expected to experience a higher intracellular Ca 2+ signal than those located at larger distances ( Meinrenken et al., 2002) (for review, see Neher, 1998). First, a differential colocalization of RRP vesicles with presynaptic Ca 2+ channels might cause a heterogeneity of p. 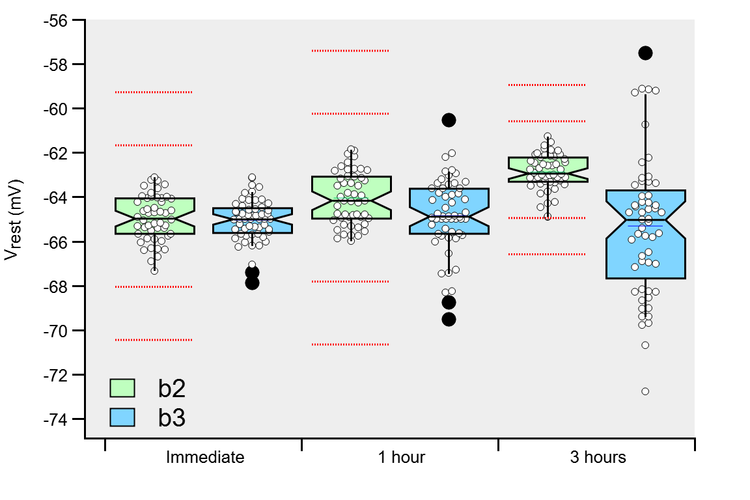
Two possible explanations immediately arise. The mechanism(s) that cause heterogeneity in release probability p between readily releasable vesicles are, however, not well understood. Heterogeneity of p is expected to strongly influence the synaptic strength during repetitive stimulation ( Trommershäuser et al., 2003) and is therefore an important factor shaping short-term plasticity of synaptic transmission. Evidence for reluctantly releasable vesicles has also been obtained at hippocampal synapses, in which an apparent increase in pool size was observed upon increasing the release probability, indicating that reluctant vesicles only contribute to transmission at elevated release probability ( Moulder and Mennerick, 2005). At the calyx of Held, a large glutamatergic synapse in the brainstem, vesicles newly recruited to the RRP have an initially low release probability ( Wu and Borst, 1999), and prolonged presynaptic voltage-clamp depolarizations lead to a fast and a slow phase of transmitter release that correspond to rapidly and more reluctantly releasable vesicles, respectively ( Sakaba and Neher, 2001a, b). In recent years, a heterogeneity of release probability has received considerable attention, because it has become apparent that some RRP vesicles are released more reluctantly than others. Thus, fast and slow transmitter release in response to prolonged i elevations is caused by intrinsic differences between RRP vesicles, and an “a posteriori” reduction of the Ca 2+ sensitivity of vesicle fusion after the onset of the stimulus might cause submaximal release of fast-releasable vesicles and contribute to short-term synaptic depression.ĭuring synaptic transmission, release of neurotransmitter is supported from a pool of docked and fusion-competent vesicles that can be defined functionally as the readily releasable pool (RRP) of vesicles ( Schikorski and Stevens, 2001 Sorensen, 2004). A model that assumes two separate subpools of RRP vesicles with different intrinsic Ca 2+ sensitivities predicted the observed Ca 2+ dependencies of fast and slow transmitter release but could not fully account for submaximal release. During trains of action potential-like presynaptic depolarizations, submaximal release was also observed as an increase in the cumulative fast release at enhanced release probabilities. 
Surprisingly, the number of vesicles released in the fast component increased with Ca 2+-uncaging stimuli of larger amplitudes, a finding that was most obvious below ∼10 μ m i and that we call “submaximal release” of fast-releasable vesicles. Ca 2+ uncaging, which produced spatially homogeneous elevations of i, evoked a fast and a slow component of release over a wide range of i, showing that mechanism(s) intrinsic to the vesicle fusion machinery cause fast and slow transmitter release. To test whether differences in the intrinsic Ca 2+ sensitivity of vesicle fusion might cause heterogeneity of p, we made presynaptic Ca 2+-uncaging measurements at the calyx of Held and analyzed the time course of transmitter release by EPSC deconvolution. Heterogeneity of release probability p between vesicles in the readily releasable pool (RRP) is expected to strongly influence the kinetics of depression at synapses, but the underlying mechanism(s) are not well understood.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
